Shock is a physiologic state characterized by a significant reduction of systemic tissue perfusion, resulting in decreased oxygen delivery to the tissues. This creates an imbalance between oxygen delivery and oxygen consumption.
Mechanism: Systemic tissue perfusion is determined by the cardiac output (CO) and systemic vascular resistance (SVR):
-
CO is the product of heart rate and stroke volume. The stroke volume is related to preload, myocardial contractility, and afterload.
-
Decreased systemic tissue perfusion is a consequence of diminished CO, SVR, or both.
Types of Shock: Four types of shock are recognized: hypovolemic, cardiogenic, distributive and obstructive.
-
Hypovolemic - Hypovolemic shock is a consequence of decreased preload due to intravascular volume loss. During hypovolemic shock, the diminished preload decreases the CO and the SVR increases in an effort to compensate for the diminished CO and maintain perfusion to the vital organs. The PCWP is decreased.
-
Cardiogenic - Cardiogenic shock is a consequence of cardiac pump failure. During cardiogenic shock, the cardiac pump failure decreases the CO and the SVR increases in an effort to compensate for the diminished CO and maintain perfusion to the vital organs. The PCWP is increased.
-
Distributive - Distributive (vasodilatory) shock is a consequence of severely decreased SVR. The CO is typically increased in an effort to compensate for the diminished SVR. The PCWP may be low or normal. Eg: sepsis, anaphylaxis, addisons crisis, toxic shock syndrome, myxedema coma, neurogenic shock, post resuscitation syndrome, post cardiopulmonary bypass.
-
Obstructive - Due to obstruction to cardiac filling or outflow like cardiac tamponade or aortic dissection, massive pulmonary embolism, tension pneumothorax, severe constrictive pericarditis, pericardial tamponade, and severe pulmonary hypertension.
Cardinal findings — Cardinal features of shock include hypotension, oliguria, abnormal mental status, metabolic acidosis, and, in some patients, cool and clammy skin.
-
Hypotension – Hypotension occurs in the majority of shock patients. It may be absolute hypotension (eg, systolic blood pressure <90 mmHg) or relative hypotension (eg, a drop in systolic blood pressure >40 mmHg). Relative hypotension explains, in part, why a patient may be in shock despite having a high or normal blood pressure. Profound hypotension may occur, with vasopressors necessary to maintain adequate perfusion pressure, as shock advances.
-
Oliguria – Oliguria may be due to shunting of renal blood flow to other vital organs, intravascular volume depletion, or both. When intravascular volume depletion is a cause, it may be accompanied by orthostatic hypotension, poor skin turgor, absent axillary sweat, or dry mucous membranes.
-
Change in mental status – The continuum of mental status changes frequently encountered in shock begins with agitation, progresses to confusion or delirium, and ends in obtundation or coma.
-
Cold, clammy skin – Potent vasoconstrictive mechanisms compensate for decreased tissue perfusion by redirecting blood from the periphery to the vital organs, thereby maintaining coronary, cerebral, and splanchnic perfusion. This causes the cold and clammy skin that is typical of shock. Not all patients with shock have cold and clammy skin though. Patients with early distributive shock or terminal shock may have flushed hyperemic skin. The former occurs prior to the onset of compensatory vasoconstriction, while the latter is due to failure of compensatory vasoconstriction.
-
Metabolic acidosis – Metabolic acidosis develops as shock progresses, reflecting decreased clearance of lactate by the liver, kidneys, and skeletal muscle. Lactate production may increase due to anaerobic metabolism if shock progresses to circulatory failure and tissue hypoxia, which can worsen the academia.
Vasopressors and Inotropic agents
Physiology of Receptors — The main categories of adrenergic receptors relevant to vasopressor activity are the alpha-1, beta-1, and beta-2 adrenergic receptors, as well as the dopamine receptors.
- Alpha adrenergic - Activation of alpha-1 adrenergic receptors, located in vascular walls, results in significant vasoconstriction.
- Beta adrenergic - Beta-1 adrenergic receptors are most common in the heart, and cause increase in inotropy and chronotropy with minimal vasoconstriction. Stimulation of beta-2 adrenergic receptors in blood vessels induces vasodilation.
- Dopamine - Dopamine receptors are present in the renal, splanchnic (mesenteric), coronary, and cerebral vascular beds; stimulation of these receptors leads to vasodilation. At higher doses between 10-20mcg/kg/min, dopamine activates alphareceptors and cause vasoconstriction. A second subtype of dopamine receptors causes vasoconstriction by inducing norepinephrine release at higher doses.
Principles of use:
The rational use of vasopressors and inotropes is guided by three fundamental concepts:
-
One drug, many receptors – A given drug often has multiple effects because of actions upon more than one receptor. For example, dobutamine increases cardiac output by beta-1 adrenergic receptor activation; however, it also acts upon beta-2 adrenergic receptors and thus induces vasodilation and can cause hypotension.
-
Dose-response curve – Many agents have dose-response curves, such that the primary adrenergic receptor subtype activated by the drug is dose-dependent. As an example, dopamine stimulates beta-1 adrenergic receptors at doses of 2 to 10 mcg/kg per minute, and alpha adrenergic receptors when doses exceed 10 mcg/kg per minute.
-
Direct versus reflex actions – A given agent can affect MAP both by direct actions on adrenergic receptors and by reflex actions triggered by the pharmacologic response. Norepinephrine-induced beta-1 adrenergic stimulation alone normally would cause tachycardia. However, the elevated MAP from norepinephrine's alpha-adrenergic receptor-induced vasoconstriction results in a reflex decrease in heart rate. The net result may be a stable or slightly reduced heart rate.
Physiological actions:
-
Norepinephrine — Norepinephrine (Levophed) acts on both alpha-1 and beta-1 adrenergic receptors, thus producing potent vasoconstriction as well as a modest increase in cardiac output. A reflex bradycardia usually occurs in response to the increased MAP, such that the mild chronotropic effect is canceled out and the heart rate remains unchanged or even decreases slightly. It also causes venoconstriction resulting in increased preload and thereby increasing cardiac output. Crit Care Med. 2012 Dec;40(12):3146-53 , Crit Care Med. 2011 Apr;39(4):689-94 , Crit Care. 2010;14(4):R142
-
Phenylephrine — Phenylephrine has purely alpha-adrenergic agonist activity and therefore results in vasoconstriction with minimal cardiac inotropy or chronotropy. MAP is augmented by raising SVR as well as increase in preload from venoconstriction. It acts predominantly by vasoconstriction of systemic capacitance veins and thereby leads to an increase in prelaod, leading to an increase in cardiac output and blood pressure. Many people think that the potential disadvantage of phenylephrine is that it may decrease stroke volume. However, studies have refuted this hypothesis and infact, phenylephrine may promote an increase in cardiac output by increasing preload in hyperdynamic septic patients. However, it does drop cardiac output in systolic heart failure. ( Please note that venoconstriction will increase the cardiac output only in patients who are preload-responsive i.e. fluid-responsive). Crit Care. 2008; 12(6): R143, Crit Care Med. 1991 Nov;19(11):1395-400 . Also, read this excellent article by Dr.Josh Farkas.
-
Epinephrine — Epinephrine (Adrenaline) has potent beta-1 adrenergic receptor activity and moderate beta-2 and alpha-1 adrenergic receptor effects. Clinically, low doses of epinephrine increase CO because of the beta-1 adrenergic receptor inotropic and chronotropic effects, while the alpha adrenergic receptor-induced vasoconstriction is often offset by the beta-2 adrenergic receptor vasodilation. The result is an increased CO, with decreased SVR and variable effects on the MAP. However, at higher epinephrine doses the alpha-adrenergic receptor effect predominates, producing increased SVR in addition to an increased CO. Epinephrine is equally potent as levophed in achieving the desired MAP. Intensive Care Med. 2008 Dec;34(12):2226-34
Other disadvantages of epinephrine include dysrhythmias and splanchnic vasoconstriction. The degree of splanchnic vasoconstriction appears to be greater with epinephrine than with equipotent doses of norepinephrine or dopamine in patients with severe shock. Epinephrine can also cause increase lactate production and can interfere with serial lactate measurements. It also decreases gut perfusion. Epinephrine can increase blood pressure in patients unresponsive to traditional agents. Read an interesting article on epinephrine hyper-responsiveness here.
-
Dopamine — Dopamine has a variety of effects depending upon the dose range administered. It is most often used as a second-line alternative to norepinephrine in patients with absolute or relative bradycardia and a low risk of tachyarrhythmias.
-
At doses of 1 to 2 mcg/kg per minute, dopamine acts predominantly on dopamine-1 receptors in the renal, mesenteric, cerebral, and coronary beds, resulting in selective vasodilation. In my opinion, these doses are useless. (also, traditionally believed to be renal protective doses).
-
At 5 to 10 mcg/kg per minute, dopamine also stimulates beta-1 adrenergic receptors and increases cardiac output, predominantly by increasing stroke volume with variable effects on heart rate.
-
At doses >10 mcg/kg per minute, the predominant effect of dopamine is to stimulate alpha-adrenergic receptors and produce vasoconstriction with an increased SVR. However, dopamine is a very weak vasopressor and causes increased mortality in sepsis. SOAP II study, Crit Care Med. 2012 Mar;40(3):725-30 , J Intensive Care Med, 27 (3) (2012), 172–178, Crit Care Med. 2015 Oct;43(10):2141-6 .
-
Dobutamine — Dobutamine is not a vasopressor but rather an inotrope that causes vasodilation. Dobutamine's predominant beta-1 adrenergic receptor effect increases inotropy and chronotropy and reduces left ventricular filling pressure. In patients with heart failure, this results in a reduction in cardiac sympathetic activity. However, minimal beta-2 adrenergic receptor effects result in vasodilation, complemented by reflex vasodilation to the increased CO. The net effect is increased CO, with decreased SVR with or without a small reduction in blood pressure.
-
Milrinone: It is a PDE inhibitor and inodilator, with both inotropic and vasodilator properties. It increases cardiac output and its effect on MAP is variable depending on the balance between inotropy and vasodilation. It decreases both PVR and SVR.
-
Isoproterenol — Isoproterenol is primarily an inotropic and chronotropic agent rather than a vasopressor. It acts upon beta-1 adrenergic receptors and, unlike dobutamine, has a prominent chronotropic effect. The drug's high affinity for the beta-2 adrenergic receptor causes vasodilation and a decrease in MAP. Therefore, its utility in hypotensive patients is limited to situations in which hypotension results from bradycardia.
-
Vasopressin: Vasopressin (antidiuretic hormone) is used in the management of diabetes insipidus and esophageal variceal bleeding; however, it may also be helpful in the management of vasodilatory shock. However, it was not shown to be any more beneficial than norepinephrine. VASST Trial
-
Methylene blue: Methylene blue increases arterial blood pressure and systemic vascular resistance in vasoplegic patients, and is especially useful in refractory shock. Critical Care Research and Practice Volume 2013 (2013), Article ID 654708 , Pharmacotherapy. 2010 Mar;30(3):323 , Crit Care Resusc. 2013 Mar;15(1):42-8. An initial dose of 2 mg/kg followed by continuous infusion of 0.25 mg/kg/hour is reasonable for less than 24 hours.
-
Levosimendan: It is a calcium sensitizer with both intotropic and vasodilator properties. In contrast to catecholamines, levosimendan causes increased myocardial contraction with a reported minimal increase in myocardial energy demand. Furthermore, levosimendan causes coronary artery vasodilation potentially improving perfusion of ischaemic myocardium. However, studies didn't show any increased benefit when compared to dobutamine. SURVIVE Trial. In LeoPARDS sepsis trial, the addition of levosimendan to standard treatment in adults with sepsis was not associated with less severe organ dysfunction or lower mortality. In CHEETAH Study, use of levosimendan for treatment of post cardiac surgery myocardial dysfunction didn't result in any difference in mortality rate, RRT or duration of mechanical ventilation.
-
Angiotensin II: Considered as an alternative to catecholamine drugs, Angiotensin II activates AT1a and AT1b receptors to increase intracellular Calcium in smooth muscle and activate the myosin contractile mechanism, thus promoting vasoconstriction. Exogenous administration of AT II augments the endogenous renin-angiotensin-aldosterone system. In ATHOS trial, Angiotensin II effectively increased blood pressure in patients with vasodilatory shock that did not respond to high doses of conventional vasopressors. However, it did not show any benefit in SOFA scores or mortality. It was recently approved by FDA at dose of 20-200 ng/kg/min. However, use it with caution as Angiotensin II is pro-thrombotic ( FDA warning for increased risk of thrombosis – 13% vs 5% in control group) and pro-inflammatory. ATHOS Trial
References: Am J Respir Crit Care Med. 2011 Apr 1;183(7):847-55 ,
Oral vasopressors:
Oral midodrine is an equivalent of phenylephrine, which is a pure alpha agonist. It reduces the duration of IV vasopressors during recovery phase from septic shock and may be associated with a reduction in length of stay in the ICU. Even though the tradional dose is 10mg TID, doses as high as 40mg TID are safely tolerated and may expedite weaning off iv vasopressors. Chest. 2016 Jun;149(6):1380-3 , J Crit Care. 2013 Oct;28(5):756-62 , J Clin Pharm Ther. 2016 Jun;41(3):260-5.
Receptors and actions
|
Norepinephrine
|
α >β1
|
2-180mcg/min
|
|
Epinephrine
|
α >β1>β2
|
2-80mcg/min
|
|
Vasopressin
|
V1R
|
0.03 units/min
|
|
Dopamine
|
0.5-2mcg/kg/min –DA
2-10 mcg/kg/min – β1 > α>DA
10-20mcg/kg/min – α >β1>DA
|
2-20mcg/kg/min
|
|
Dobutamine
|
β1>β2
|
2-20mcg/kg/min
|
|
Isoproterenol
|
β1=β2
|
|
|
Milrinone
|
PDE 3 Inhibitor
|
0.25-0.75 mcg/kg/min
|
|
Phenylephrine
|
α
|
20-500mcg/min
|

Side Effects of catecholamine vasopressors:
-
Excess levels of catecholamines is associated with stress cardiomyopathy
-
Catecholamines through beta receptors causes inhibition of insulin release and stimulation of catabolic hormones like cortisol and glucagon. Hence, it causes more lipolysis and glycogenolysis.
-
Stimulate a prothrombotic tendency and inhibit fibrinolytic system
-
Suppress neutrophil function
-
Can increase bacterial growth by increasing bacterial uptake of iron, which is so essential for bacterial multiplication.
Vasopressors from peripheral IV :
Very frequently we think that vasopressors need central lines because we were told so by our peers and teachers. I often see that people think its safe to administer dopamine through peripheral IV but unsafe to administer levophed , phenylepnrine or vasopressin, even though each of these medications is trying to achieve the same degree of vasoconstriction to raise the MAP. Extravasation occurs in less than 2% of patients and even in those, the incidence of limb threatening ischemia is virtually non existent. Even if the drug gets infiltrated, could one agent be worse than other if they are causing the same degree of vasoconstriction ?
If you think of it, epinephrine upto 1 mg ( 1:1000 concentration or 1000 mcg/ml) is approved for intramuscular or SQ injection in case of anaphylaxis and 1 mg ( 1:10000 concentration or 100mcg/ml) as iv push for cardiac arrest. These are much more concentrated forms than the epinephrine drips that we run ( typically 40mcg/ml ). Also, phenylephrine has been used safely for decades as SQ injections upto a dose of 10,000 mcg. If concentrated epinephrine and phenylephrine are safe for subcutaneous injection, then extravasation of diluted infusions should be tolerated. Also, epinephrine works as its own antidote as it has both alpha action and beta 2 action with resultant vasodilation.
Numerous studies have showed that administration of vasopressors through peripheral IV ( atleast 20G with blood return) is completely feasible and safe to do so for a limited period of time. Extravasation from the peripheral intravenous line was uncommon, and phentolamine with nitroglycerin paste were effective in preventing local ischemic injury. In case of extravasation, phentolamine in a dose of 0.1 to 0.2 mg/kg (up to a maximum of 10 mg) should then be injected through the catheter and subcutaneously around the site. Clinicians should not completely disregard the feasibility of vasoactive medications through a peripheral iv access. J Hosp Med. 2015 Sep;10(9):581-5 , Ann Emerg Med. 2006 Jun;47(6):559-63 , J Crit Care. 2016 Aug;34:107-10 , J Crit Care. 2015 Jun;30(3):653.e9-17. Not only administration of vasopressors is helpful in avoiding unnecessary central lines but also avoid giving unnecessary fluid boluses with a goal of stabilising blood pressure.
My take home message: Its safe to use vasopressors through a good 20G peripheral IV for a period less than 48 hours from the same IV and as long as the requirements remain less than 20 mcg of norepinephrine or 30 mcg of epinephrine or 100mcg of phenylephrine or 0.04units/min of vasopressin. Also, epinephrine is much safer than norepinephrine through peripheral iv due to its beta 2 action with potential dermal vasodilation. One way to minimise the risk, if extravasated, is to dilute the medication in more normal saline or D5W. Read this excellent article by Dr.Josh Farkas and this article.
Crystalloids and colloids in shock:
Crystalloids (IV fluids) in shock:
Even though most of us do not even think iv fluids as medications, IV fluids are one of the most dangerous drugs in the ICU, when used inappropriately.
Stressed and Unstressed volume:
The blood volume may be partitioned into stressed volume, which contributes directly to venous return, and unstressed volume, which does not but instead is stored mainly in large capacitance veins. In normal conditions, a significant part of the blood constitutes unstressed volume. In compensated hypovolaemia , unstressed volume is decreased but stressed volume, and hence venous return and cardiac output , is preserved. In uncompensated hypovolaemia, both unstressed and stressed volumes are decreased, and venous return and cardiac output cannot be maintained. In sepsis, vasodilation increases the volume in which blood distributes (the size of the ‘tank’), which may be termed relative hypovolaemia. Hence, unstressed volume is increased leading to decreased stressed volume, and venous return and cardiac output cannot be preserved even though total blood volume is not decreased.
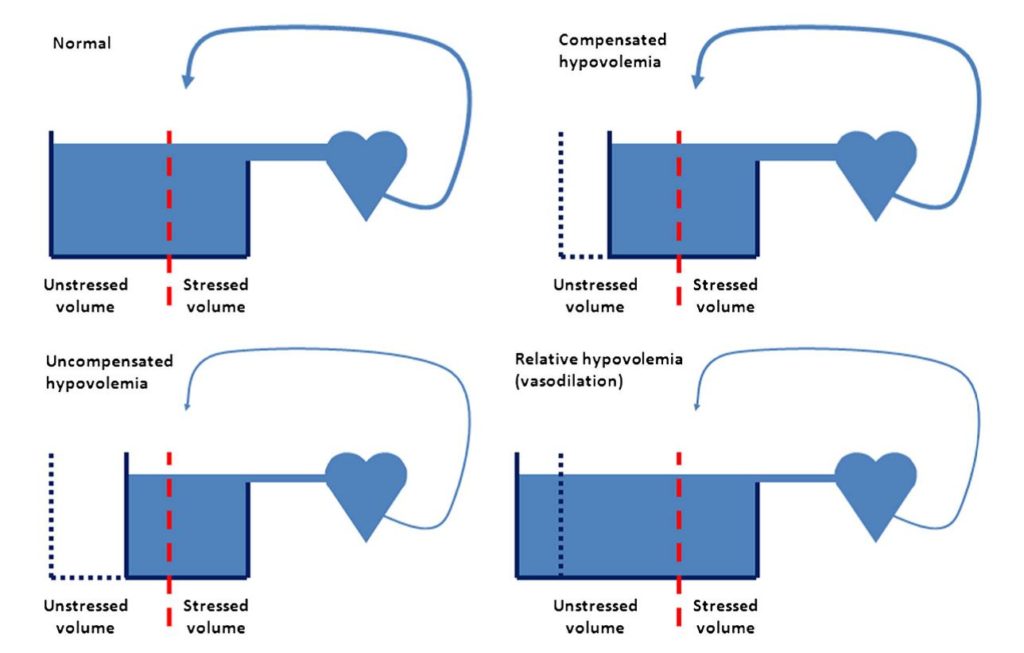
When we assess a shocked patient we often start interventions based on limited information. The first question we have to answer is ‘Will my patient benefit from increased cardiac output?’ If yes, then markers of hypovolaemia should be assessed. Several dynamic indices are available, which are discussed in hemodynamics chapter. The fluid bolus should increase preload measured as central venous pressure or caval vein diameter. If not, the volume given may not have increased the stressed volume to challenge cardiac function. Arterial blood pressure changes during fluid challenge, on the other hand, are of less value, because they are poor markers of changes in cardiac output unless the pressure changes are marked.
The circulatory effects of fluid resuscitation are determined by the interaction of venous return and cardiac function. Central to the understanding of the venous return function is the mean systemic filling pressure , which drives the gradient for venous return and is determined by the amount of volume in the circulation and the compliance of the capacitance vessels. Venous return enters the heart depending on the gradient between mean systemic feeling pressure and right atrial pressure. Cardiac function regulates right atrial pressure, thereby enabling venous return and filling of the heart in diastole. In order to understand when to use fluids and when not to use fluids, an understanding of venous return and cardiac output is of paramount importance. Intensive Care Med. 2014 Oct;40(10):1564-6 , Intensive Care Med. 2014 Apr;40(4):613-5
Most of us were taught that sepsis resuscitation should start with volume resuscitation before initiating vasopressors (“a.k.a fill the tank first”). It is also a very common practice to load septic patients with a certain arbitrary amount of volume (30cc/kg) before starting vasopressors. The theory behind this is that septic patients are often intravascularly volume depleted due to third-space losses, and so arterial constriction alone could impair perfusion. However, studies have failed to prove this. Crit Care. 2012 Jan 25;16(1):302 , Crit Care. 2011;15(3):164 Infact, studies showed that early vasopressors not only prevent fluid overload but also improve cardiac output and perfusion , thereby limiting mortality and organ damage.
A lot of us commonly use iv fluids as a replacement for vasopressors resulting in significantly positive fluid balance in 48-72 hours, sometimes as much as 10 litres. Also, after the patient was intially resuscitated with iv fluids on admission and establish a normovolemia, there is no need for additional fluids unless there are ongoing volume losses. I fail to understand why clinicians give more fluids when the patient is already in 10 litres positive fluid balance, even when the patient is grossly edematous with pulmonary congestion. Clearly, that patient is not hypovolemic anymore and definitely doesn't need additional volume. For example, a patient on the floor with a pneumonia, day3, becomes hypotensive due to sepsis and he would be bombarded with 4-5 litres of iv fluids before starting vasopressors. This method is very flawed and pose many dangers to the patient.
The traditional thinking was to never start vasopressors without adequate volume ( in common words, without filling the tank). Even though it was true to a certain extent, starting vasopressors early, even while the fluid resuscitation is going on, showed a 3% relative risk reduction in mortality for each hour of vasopressors started early. Also, please remember that once the patient is volume replete ( most of which happens in the ED itself, thanks to the blind protocols which recommend 30cc/kg for everyone ) , they don't need additional volume. If they are in vasodilatory shock, they need vasopressors. Please do not treat vasodilatory shock as hypovolemic shock. Critical Care 2013, 17(Suppl 1):S6. As Paul Marik described, early management of sepsis doesn't include only iv fluids and antibiotics. Chest. 2014 Jun;145(6):1407-18. Another interesting article here by Dr.Farkas.
Also, in patients who are volume depleted, iv fluids should always be given as boluses rather than as maintainence iv fluids, unless there are ongoing fluid losses. Various bedside dynamic indices should be used to decide on giving additional iv fluids. These dynamic indices include passive leg raising test, IVC collapsibility or distensibility, Pulse pressure variation, stroke volume variation, mini volume challenges, changes in CVP with spontaneous breathing, and change in VTI with a mini bolus. Empty IVC + hyperkinetic heart does not equal volume depletion, as described beautifully here.
Colloids ( Albumin) in shock:
The primary rationale for administering colloid solution in the setting of hypovolemia is the desire to expand plasma volume more effectively. The larger solutes present in colloidal solutions are theoretically resistant to passage across the capillary membrane, and hence preserve intravascular oncotic pressure and prevent extravasation in accordance with the Starling equation. While this may work in theory, randomized controlled human trials comparing crystalloids with colloids suggest no difference. SAFE Trial, ALBIOS Study. Infact, colloids like hexastarch have been shown to cause increased mortality , deranged hemostasis and increased organ dysfunction. N Engl J Med 2012; 367:124-134. Also, colloids has been shown to increase kidney dysfunction when compared to crystalloids. Crit Care Med. 2011 Jun;39(6):1335-42 , Intensive Care Med. 2008 Dec;34(12):2157-68.
Important adverse effects of over resuscitation and positive fluid balance are:
-
Positive fluid balance is associated with worse mortality. Intensive Care Med. 2017 Jan 27 , Crit Care. 2014; 18(Suppl 2): P70 , Crit Care. 2015; 19(1): 251 , Indian J Crit Care Med. 2015 Dec; 19(12): 708–713.
- Abdominal compartment syndrome
-
Chloride rich fluids can cause AKI. JAMA. 2012 Oct 17;308(15):1566-72
-
Chloride rich fluids can damage endothelial glycocalyx and cause more capillary leak, resulting in a vicious cycle. Pflugers Arch. 2011 Oct;462(4):519-28.
PEARLS:
-
Normal saline is the most common fluid used in ICU's . But, there is nothing normal about normal saline, which is the most common iv fluid used in ICU. It was originally invented to replace fluid losses in cholera patients, who lose chloride rich colonic fluid. It is severely hyperchloremic, severely acidotic with a PH of 5.6 and can cause kidney damage. Lancet. 1989 Apr 8;1(8641):768-71, Int J Med Sci. 2013; 10(6): 747–750
-
Bradycardia: Use dopamine if hypotensive and Isoproterenol if normotensive.
-
Epinephrine has equal inotropic effect as dopamine and much more than norepinephrine.
-
Norepinephrine and epinephrine produce similar splanchnic blood flow in moderate shock but Epinephrine causes splanchnic vasoconstriction in severe shock.
-
Non survivors demonstrated extremely high levels of catecholamines in body, >4000pg/ml of epinephrine and >10,000pg/ml of norepinephrine. Normal levels are <200pg/ml of epinephrine and <500pg/ml of norepinephrine. Total volume of plasma is around 3litres. Hence, normal total value of epinephrine is 0.6mcg and norepinephrine is 1.5mcg.
-
Standard dose for vasopressin is 2 units/hr. However, vasopressin may be given in doses up to 20units/hr. Infact, vasopressin dose in ACLS is 40units. In some of the old studies, vasopressin doses as high as 2 units/min have been used.
-
Each hour delay with inadequate MAP increases the risk of AKI. So, never allow lower MAP's for too long. Even while the fluid boluses are running, give push dose pressors to maintain adequate MAP at all times. Comput Cardiol (2010). 2010;37:1095-1098
-
Very nicely written article.
Ever thought of Non obstructive mesenteric ischaemia in cases of septic shock.Norepinephrine would be disaster
Any evidence that you can offer to our audience so we are better informed. Appreciate the input.
I have a question. I am a PT in the ICU. I often hear the phrase the patient is maxed out on pressors. Some nurses have told me three is max some four. I would like some insight as to what is the max? My exclusion criters states for the patient to be excluded from my early mobility program if they "are requiring a significant dose of vasopressors". Im trying to understand in detail what that means. Thank you.
There is no theoritical max dose of pressors. However, each vasopressor acts on their specific receptors and traditional thinking was that there are only so many receptors for those drugs to act on. One we saturate those receptors with a particular vasopressor, increasing the dose will not add any value. We can add different vasopressors with different mechanism of action and targetting different receptors. Still, there is no limit on the number of vasopressors that you can use. However, if patient is not responding to 3 or more vasopressors, it may not even be the vasodilation issue and we need to dig deeper. For example, a tamponade or massive PE will need a different strategy than vasopressors alone.
Any mobility exercises will put lot more stress on heart and will possibly require more vasopressors. The dose above which mobility is contraindicated is subject to debate. Personally, i am okay with patients moving in the ICU with less than 5mcg/min of levophed. Any dose of vasopressor is acceptable if its just to get them to a chair.